When he states that something is impossible, he is very probably wrong.” Left to right: Robert Millikan and Albert Einstein in 1932, important protagonists in the development of atomic theory Ernest Rutherford, who developed the modern conception of the atom describing the nucleus James Chadwick, discoverer of the neutron in 1932 under Rutherford's mentorship Science fiction author H.G. Clarke’s famous laws of scientific research: “When a distinguished but elderly scientist states that something is possible, he is almost certainly right. These uncharacteristically myopic statements bring to mind one of Arthur C. 
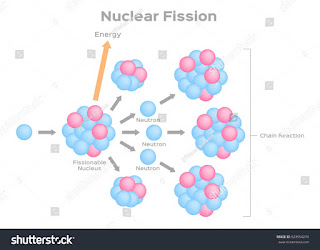
“There is no likelihood that man can ever tap the power of the atom… The glib supposition of utilizing atomic energy when our coal has run out is a completely unscientific utopian dream.” - Robert Millikan, 1928. “Anyone who expects a source of power from these atoms is talking moonshine.” - Ernest Rutherford, 1933. It would mean that the atom would have to be shattered at will.” - Albert Einstein, 1934. “There is not the slightest indication that will ever be obtainable. Surprisingly, in only the decade before, some of the greatest scientists of the era, including ones that were directly responsible for the creation of the field, still held deeply pessimistic views on the prospect of nuclear fission: The discovery of plutonium is inexorably entwined with the development of nuclear fission, i.e., the splitting of the atom. The ink outlining the concept of a top-secret uranium bomb was still wet on the page, but in this tangential effort, they had almost immediately found a superior element for its fissionable component. Yet the details of this momentous discovery weren't shared with the rest of the scientific community until 1946, after World War II-the only time a fundamental scientific discovery of this importance has ever been kept secret. Discovery: Hesperium, Extremium, or Plutonium?Įlement 94 was first isolated in minute quantities in 1941 by Glenn Seaborg, Edwin McMillan, Joseph Kennedy, and Arthur Wahl at the Radiation Laboratory of the University of California, Berkeley. In this article, I attempt to shine a light on this fascinating element with particular focus on the unique story of its discovery and its strange properties that continue to challenge our best theories. As such, the study of plutonium is an inherently interdisciplinary activity. These properties are, however, almost entirely disconnected from the nuclear properties which plutonium is more widely valued for. At a basic level, this is because of its location in the periodic table, at a tipping point between localized and delocalized electronic configurations. 
It actually increases in density when it melts and it is a poor conductor of heat or electricity-and the list of anomalies goes on and on. From the orbits of an astronomical scale to those of the subatomic, we have also gained an incredible opportunity to peer into the deep complexities of the atom and the myriad competing factors that determine its properties, many of which are still a mystery to us.Īlbert Migliori, former director of the Seaborg Institute in Los Alamos has said that “it is not unreasonable to consider plutonium as the most interesting element after helium in its challenge to our understanding.” An element that has been called a physicist’s dream but an engineer’s nightmare, it can change density by as much as 25% with little provocation. Despite being an element that was born in wartime, and controversially used to help end that conflict, it has also found peaceful use as a source of both nuclear and thermoelectric power, in turn enabling the exploration of the solar system. While this reputation is surely justified in part, it is but one aspect of this anarchically temporal and almost paradoxical metal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
